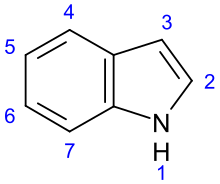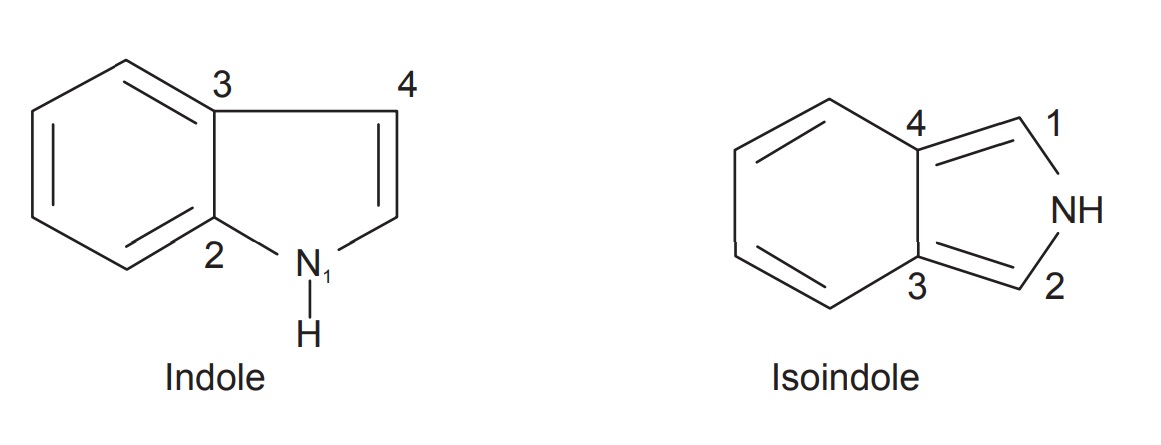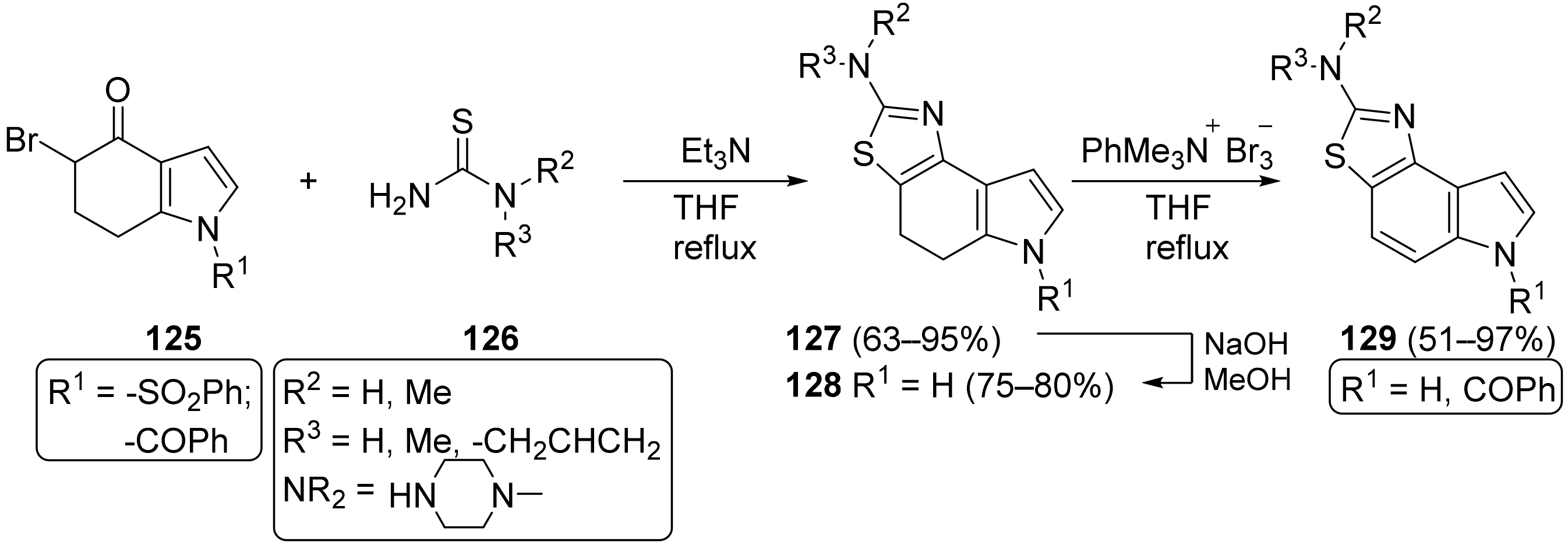
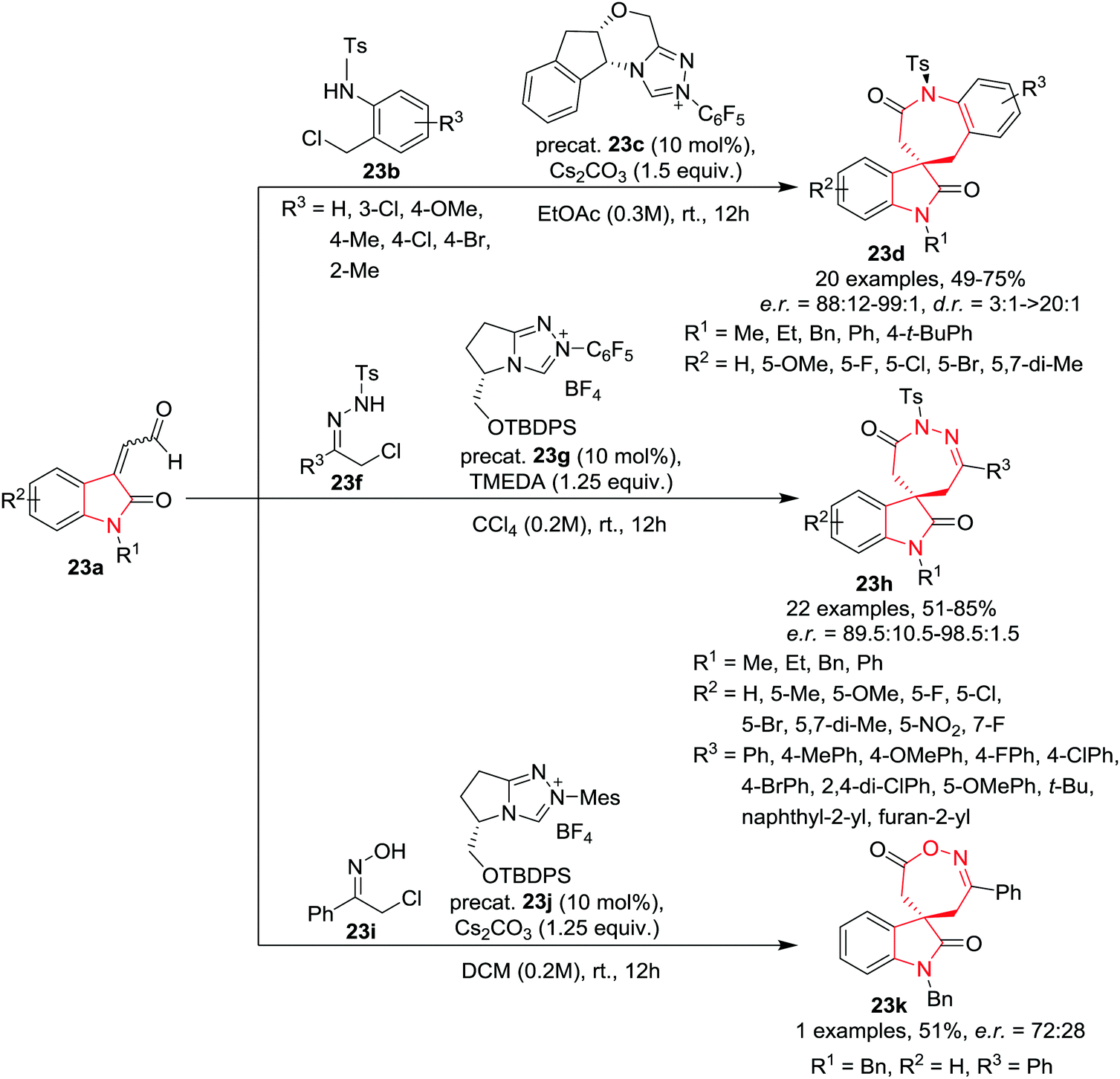
Molecules | Free Full-Text | 4,5,6,7-Tetrahydroindol-4-Ones as a Valuable Starting Point for the Synthesis of Polyheterocyclic Structures | HTML

Indole decreases the virulence of the bivalve model pathogens Vibrio tasmaniensis LGP32 and Vibrio crassostreae J2-9 | Scientific Reports
Recent advances in spirocyclization of indole derivatives - Chemical Society Reviews (RSC Publishing) DOI:10.1039/C7CS00508C

Generic structure of indole-based synthetic cannabinoids (adapted from... | Download Scientific Diagram
![Synthesis of the Azocino[cd]indole Framework through Pd‐Catalyzed Intramolecular Acetylene Hydroarylation - Peshkov - 2011 - European Journal of Organic Chemistry - Wiley Online Library Synthesis of the Azocino[cd]indole Framework through Pd‐Catalyzed Intramolecular Acetylene Hydroarylation - Peshkov - 2011 - European Journal of Organic Chemistry - Wiley Online Library](https://chemistry-europe.onlinelibrary.wiley.com/cms/asset/64d0fce9-3360-4b5e-94c6-97bb8fa6167d/mfig000.jpg)
Synthesis of the Azocino[cd]indole Framework through Pd‐Catalyzed Intramolecular Acetylene Hydroarylation - Peshkov - 2011 - European Journal of Organic Chemistry - Wiley Online Library

Tandem Nenitzescu Reaction/Nucleophilic Aromatic Substitution to Form Novel Pyrido Fused Indole Frameworks - Singh - 2021 - European Journal of Organic Chemistry - Wiley Online Library

Design, synthesis, antitubercular and antiviral properties of new spirocyclic indole derivatives | SpringerLink

Indole decreases the virulence of the bivalve model pathogens Vibrio tasmaniensis LGP32 and Vibrio crassostreae J2-9 | Scientific Reports

Synthetic Methods for 3,4‐Fused Tricyclic Indoles via Indole Ring Formation - Nemoto - 2018 - Asian Journal of Organic Chemistry - Wiley Online Library

Indole decreases the virulence of the bivalve model pathogens Vibrio tasmaniensis LGP32 and Vibrio crassostreae J2-9 | Scientific Reports









![Tryptophan [Natural Amino Acid Derivatives] | TCI EUROPE N.V. Tryptophan [Natural Amino Acid Derivatives] | TCI EUROPE N.V.](https://www.tcichemicals.com/medias/T0540.jpg?context=bWFzdGVyfHJvb3R8MzEwNzJ8aW1hZ2UvanBlZ3xoMjMvaDA0Lzg5MzIzNjA3MTYzMTgvVDA1NDAuanBnfGQ5ZmZhZTAwZjcyNDdhODFkMjFiNWY3ZjZiM2NjZWI3NTU3NjAxMGU1NTNjOTNlYzk1YjRiNjExYzhkNmJhOTE)