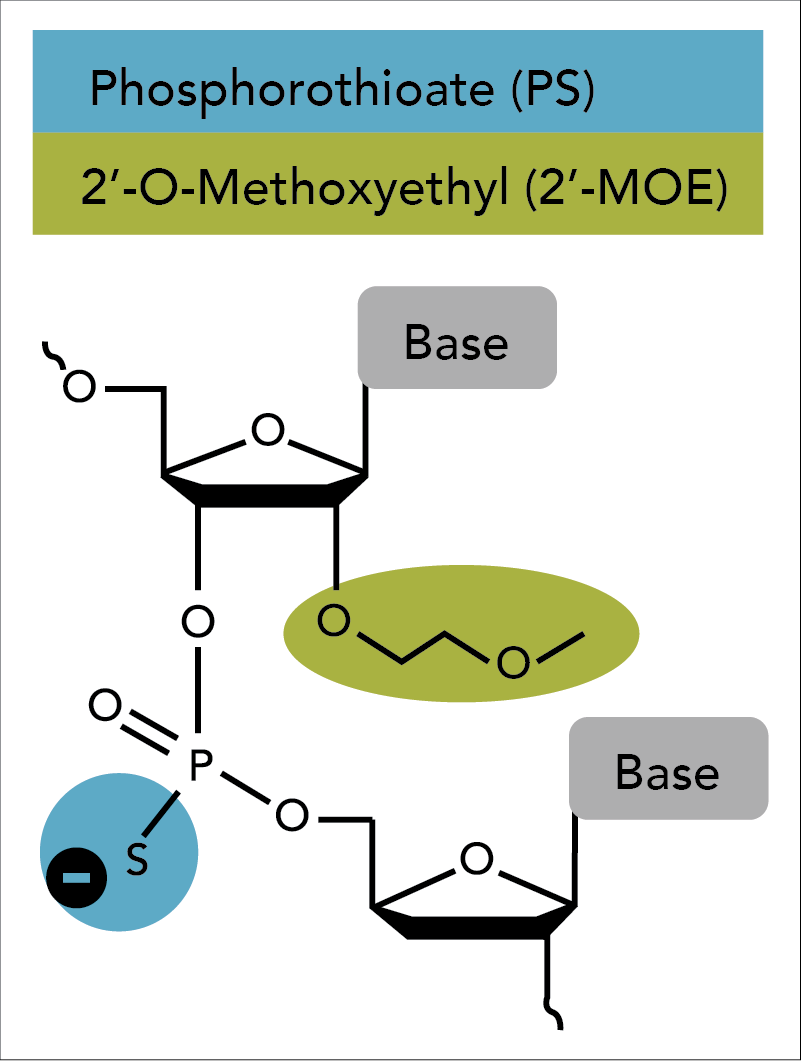
An ASO modification that enhances nuclease resistance, lowers toxicity, and increases binding affinity

DUX4 Transcript Knockdown with Antisense 2′-O-Methoxyethyl Gapmers for the Treatment of Facioscapulohumeral Muscular Dystrophy - ScienceDirect

Oligonucleotide therapeutics: How to measure plasma concentration, tissue distribution and immunogenicity?

Safety and efficacy of olesoxime in patients with type 2 or non-ambulatory type 3 spinal muscular atrophy: a randomised, double-blind, placebo-controlled phase 2 trial - The Lancet Neurology

Understanding European patient expectations towards current therapeutic development in spinal muscular atrophy - Neuromuscular Disorders
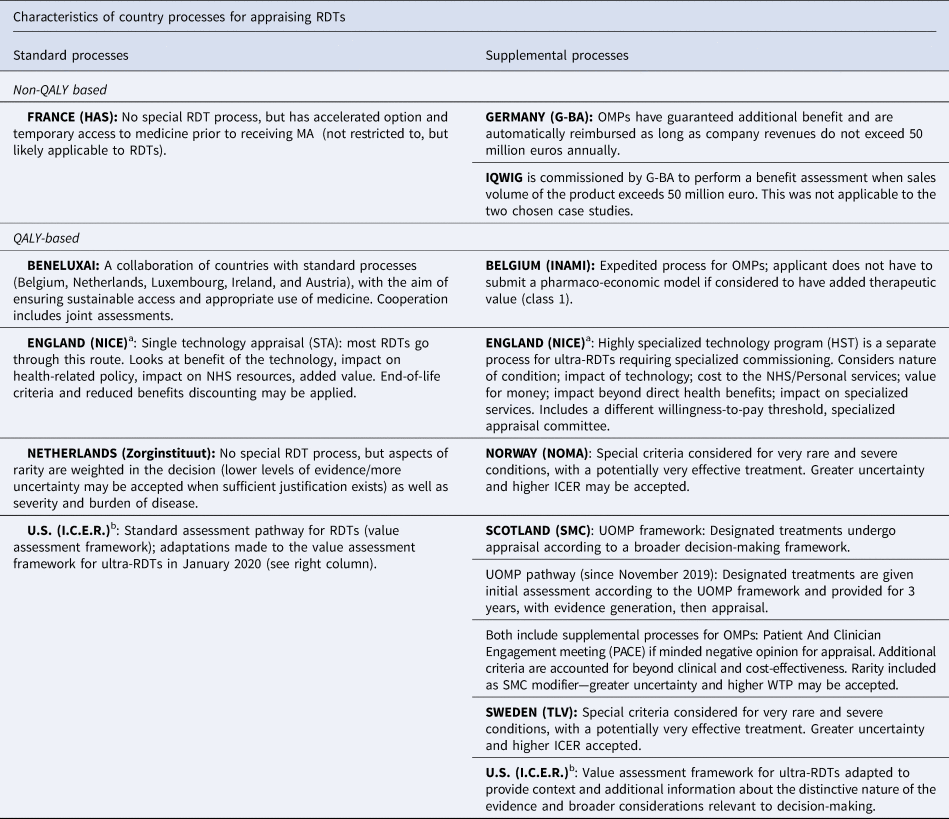
Examining the impact of different country processes for appraising rare disease treatments: a case study analysis | International Journal of Technology Assessment in Health Care | Cambridge Core
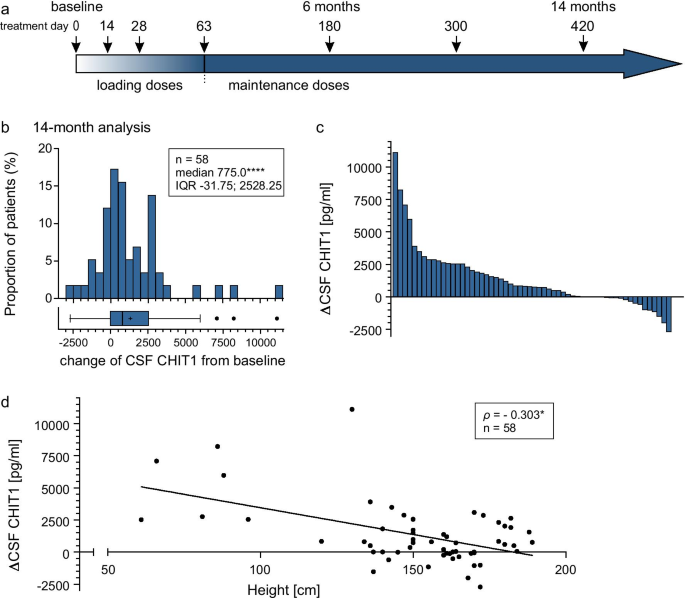
Increased chitotriosidase 1 concentration following nusinersen treatment in spinal muscular atrophy | Orphanet Journal of Rare Diseases | Full Text
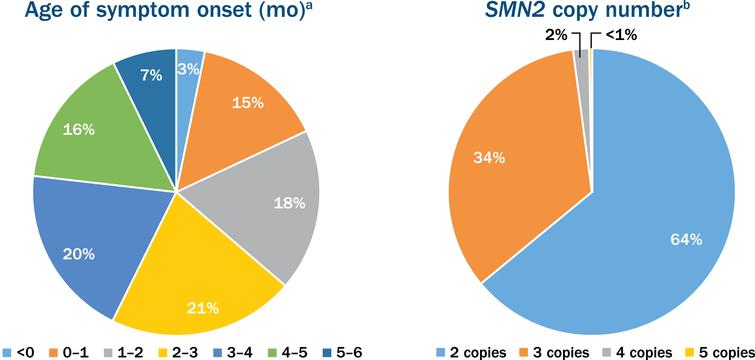
Implementing a Global Expanded Access Program (EAP) for Infantile-Onset Spinal Muscular Atrophy (Type I): Understanding the Imperative, Impact and Challenges - IOS Press

Nusinersen treatment significantly improves hand grip strength, hand motor function and MRC sum scores in adult patients with spinal muscular atrophy types 3 and 4 | Request PDF
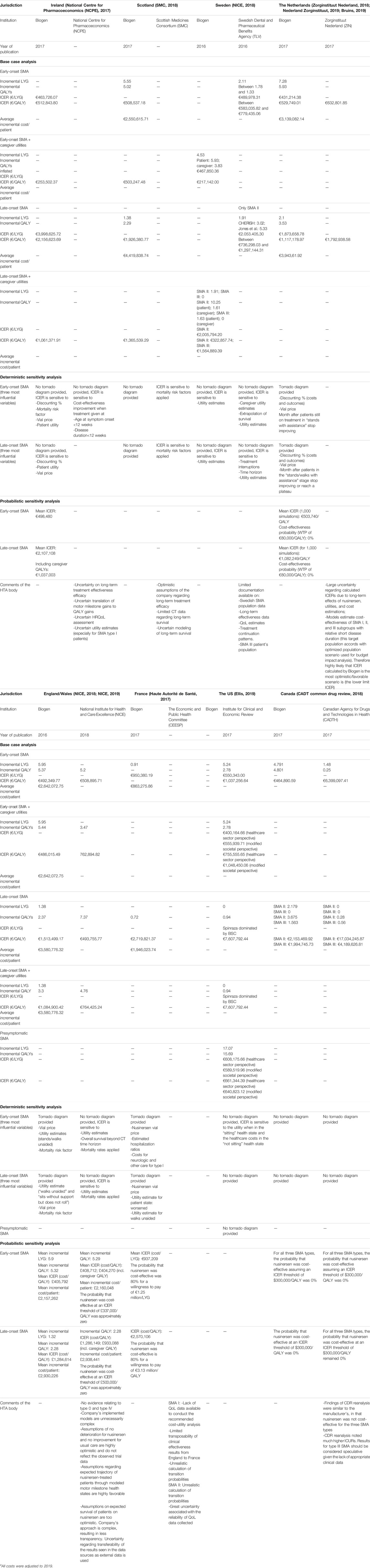
Frontiers | Assessing the Value of Nusinersen for Spinal Muscular Atrophy: A Comparative Analysis of Reimbursement Submission and Appraisal in European Countries | Pharmacology
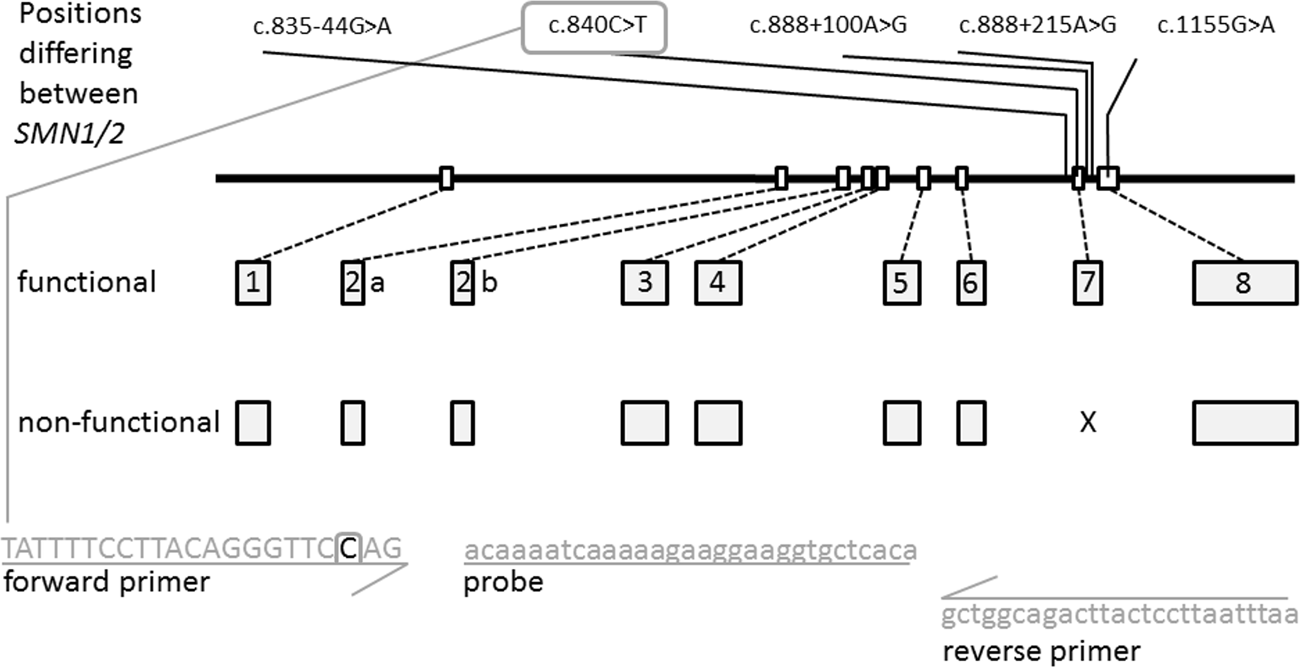
High-throughput genetic newborn screening for spinal muscular atrophy by rapid nucleic acid extraction from dried blood spots and 384-well qPCR | European Journal of Human Genetics
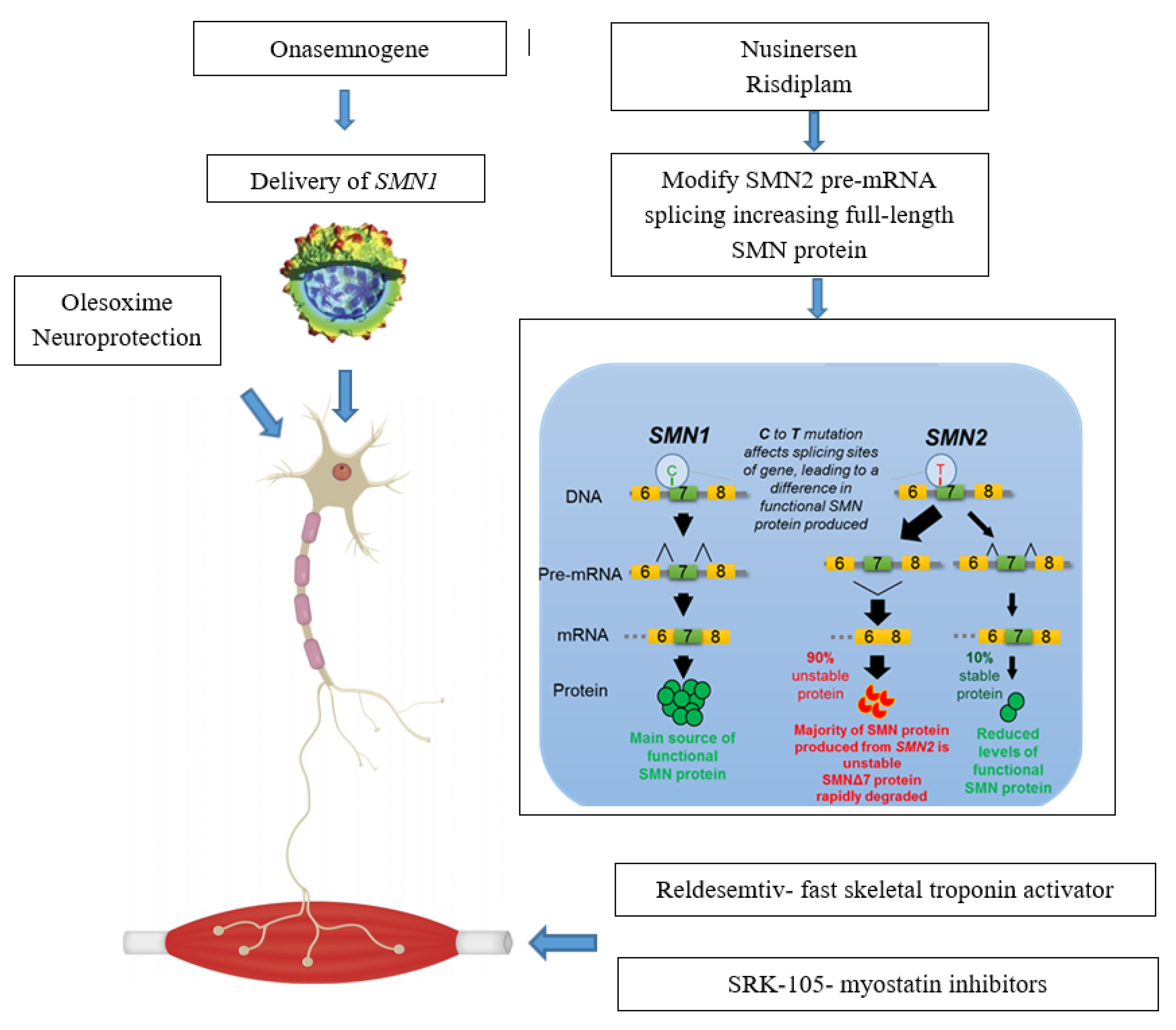
JCM | Free Full-Text | New Treatments in Spinal Muscular Atrophy: Positive Results and New Challenges | HTML

Constantin d'Ydewalle - Senior Scientist, Neuroscience Drug Discovery - The Janssen Pharmaceutical Companies of Johnson & Johnson | LinkedIn
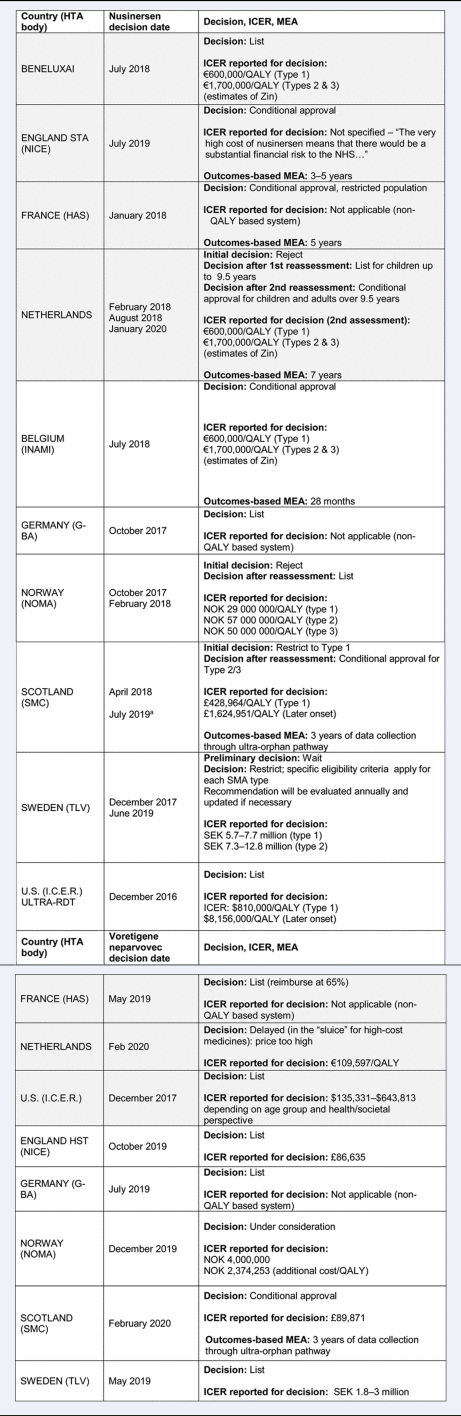
Examining the impact of different country processes for appraising rare disease treatments: a case study analysis | International Journal of Technology Assessment in Health Care | Cambridge Core
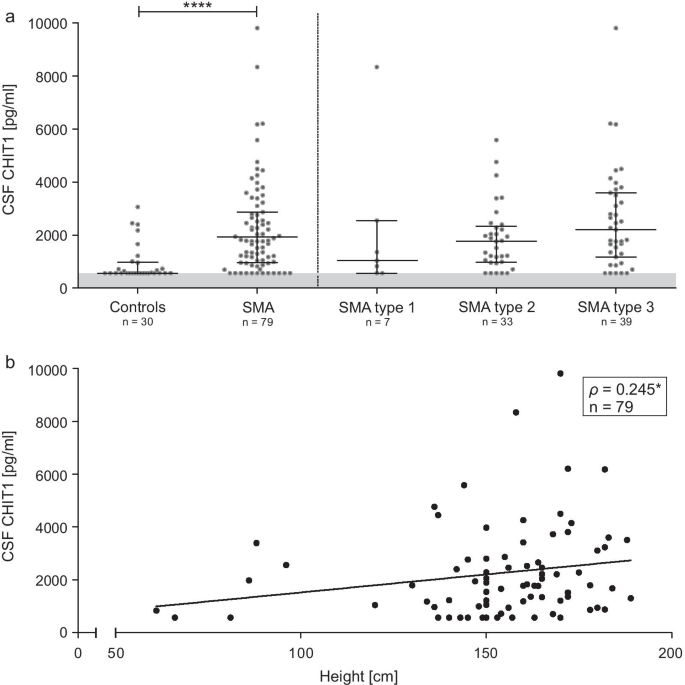
Increased chitotriosidase 1 concentration following nusinersen treatment in spinal muscular atrophy | Orphanet Journal of Rare Diseases | Full Text
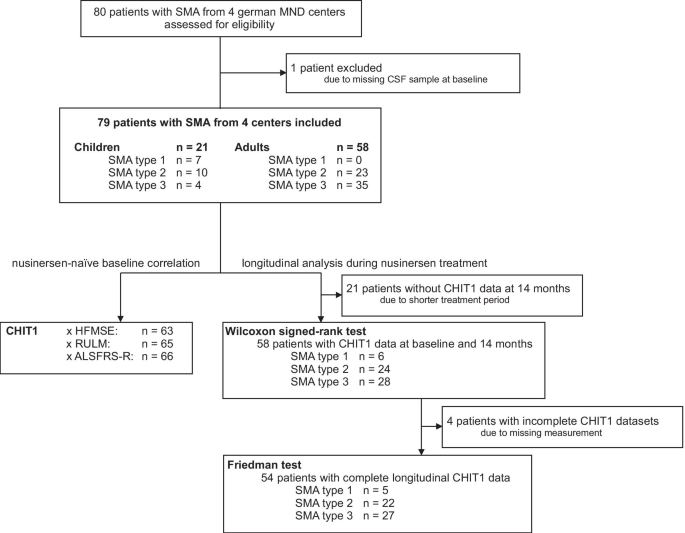
Increased chitotriosidase 1 concentration following nusinersen treatment in spinal muscular atrophy | Orphanet Journal of Rare Diseases | Full Text