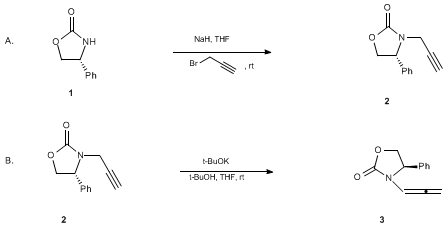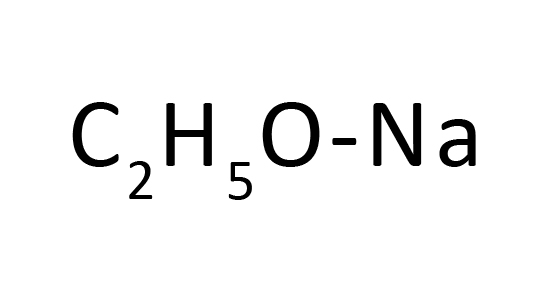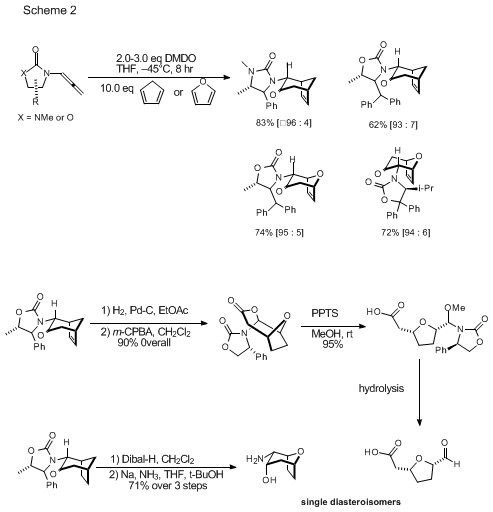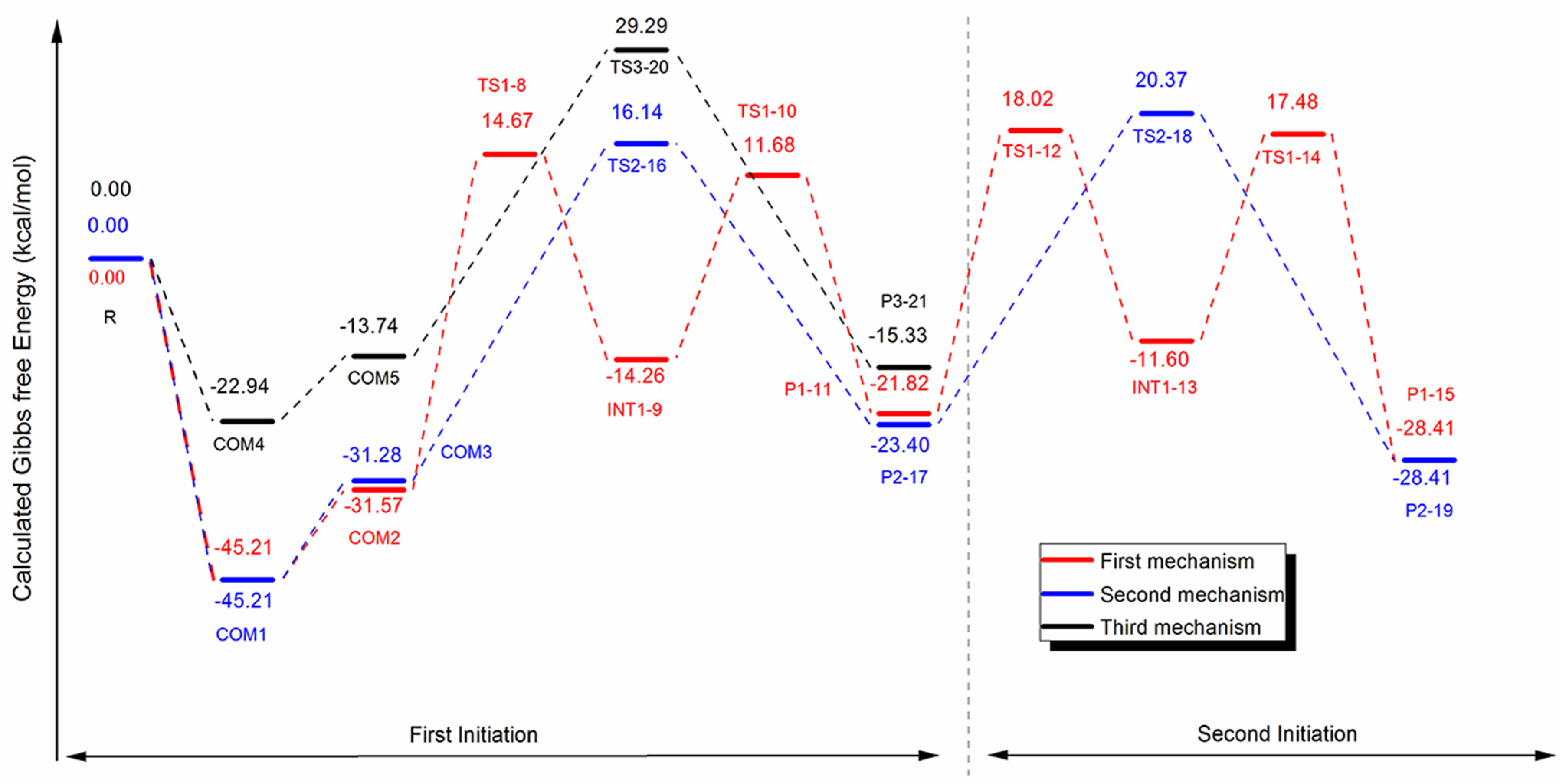
Polymers | Free Full-Text | Organocatalytic Ring-Opening Polymerization of ε-Caprolactone Using bis(N-(N′-butylimidazolium)alkane Dicationic Ionic Liquids as the Metal-Free Catalysts: Polymer Synthesis, Kinetics and DFT Mechanistic Study ...

Aerobic oxidation of secondary benzylic alcohols and direct oxidative amidation of aryl aldehydes promoted by sodium hydride - ScienceDirect

Understanding the Origins of Nucleophilic Hydride Reactivity of a Sodium Hydride–Iodide Composite - Hong - 2016 - Chemistry – A European Journal - Wiley Online Library

The Employment of Sodium Hydride as a Michael Donor in Palladium‐catalyzed Reductions of α, β‐Unsaturated Carbonyl Compounds - Liu - 2019 - Advanced Synthesis & Catalysis - Wiley Online Library

An unexpected reaction to methodology: an unprecedented approach to transamidation - RSC Advances (RSC Publishing) DOI:10.1039/C6RA23374K

Amide‐Directed C−H Sodiation by a Sodium Hydride/Iodide Composite - Huang - 2017 - Angewandte Chemie International Edition - Wiley Online Library
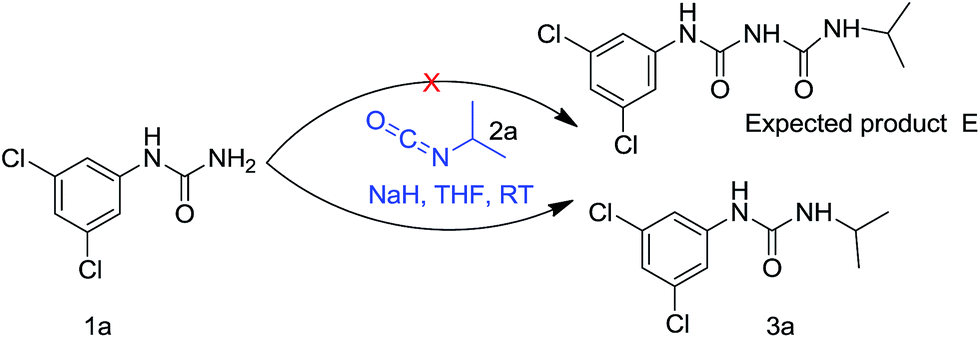
An unexpected reaction to methodology: an unprecedented approach to transamidation - RSC Advances (RSC Publishing) DOI:10.1039/C6RA23374K

Nucleophilic amination of methoxypyridines by a sodium hydride–iodide composite - Chemical Communications (RSC Publishing) DOI:10.1039/C8CC05979A

Sodium Hydride Catalyzed Tishchenko Reaction - Werner - 2010 - European Journal of Organic Chemistry - Wiley Online Library