
A comparative study of N‐hydroxylating flavoprotein monooxygenases reveals differences in kinetics and cofactor binding - Ernst - - The FEBS Journal - Wiley Online Library
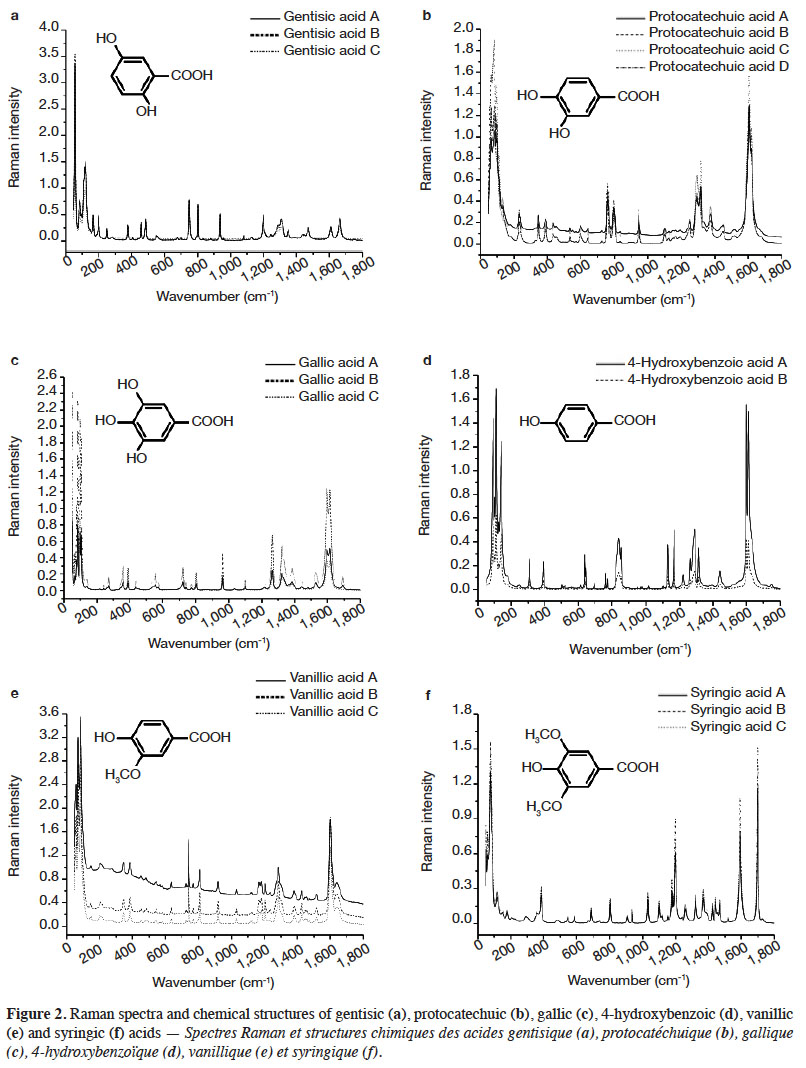
Characterization and discrimination of phenolic compounds using Fourier transform Raman spectroscopy and chemometric tools | Université de Liège
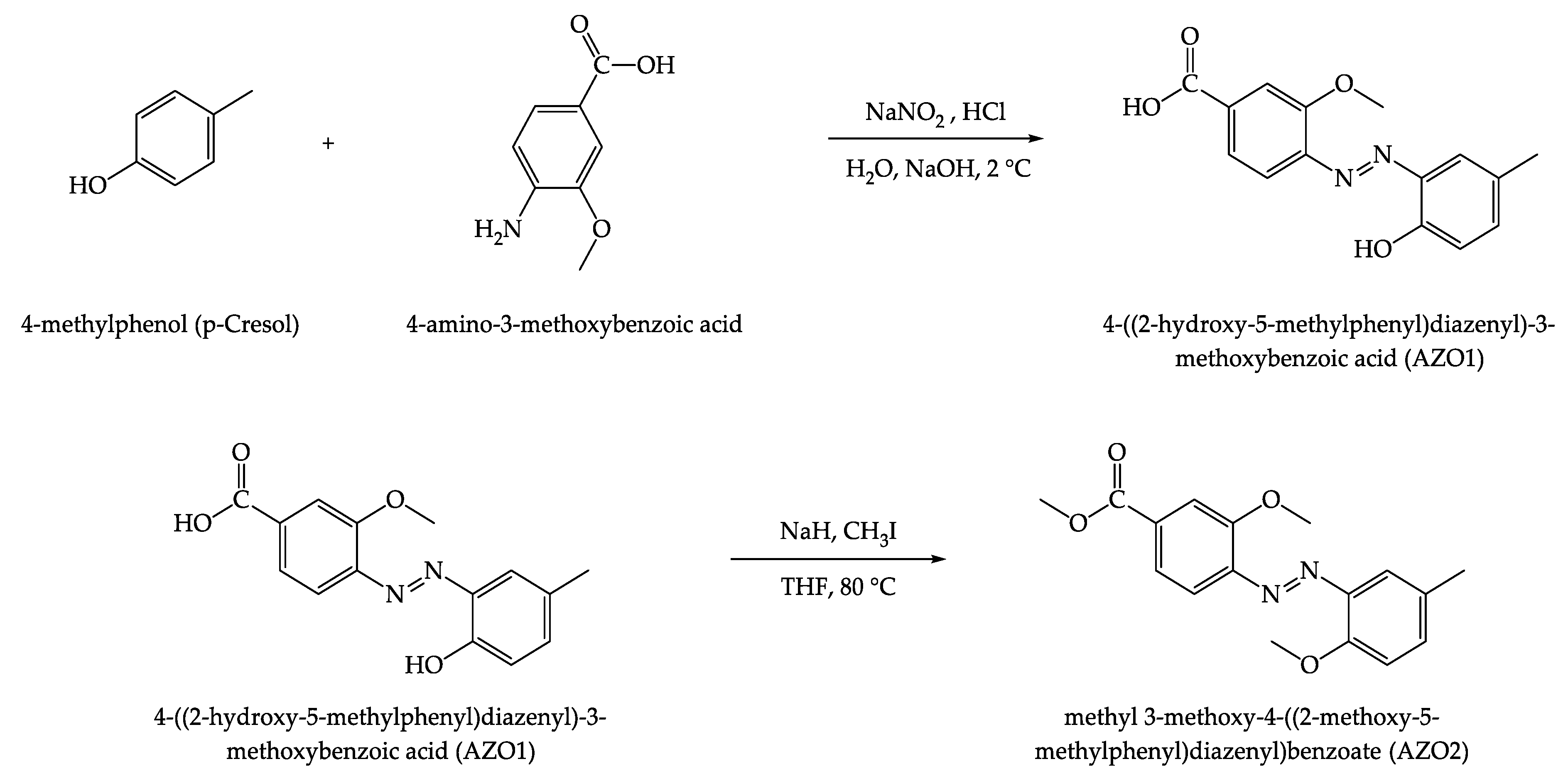
Polymers | Free Full-Text | Contrasting Photo-Switching Rates in Azobenzene Derivatives: How the Nature of the Substituent Plays a Role | HTML
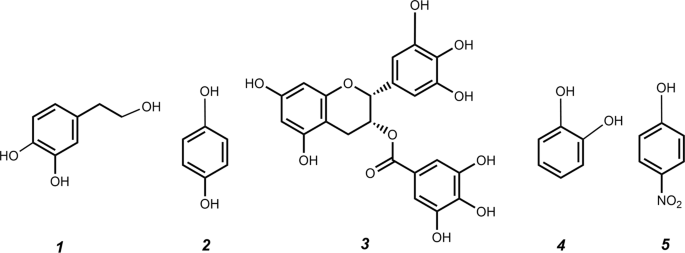
Deciphering the molecular specificity of phenolic compounds as inhibitors or glycosyl acceptors of β-fructofuranosidase from Xanthophyllomyces dendrorhous | Scientific Reports
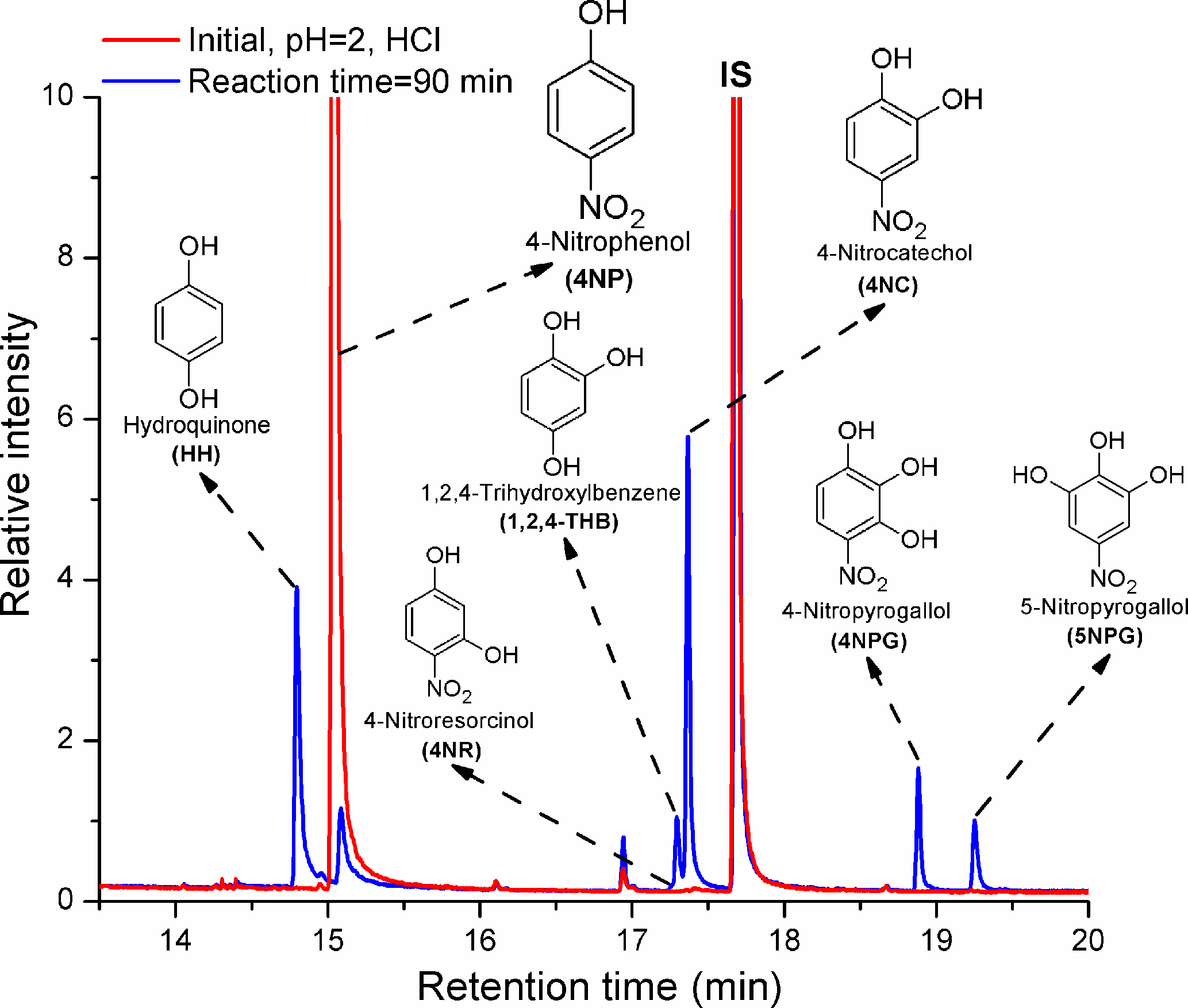
ACP - Aqueous chemical bleaching of 4-nitrophenol brown carbon by hydroxyl radicals; products, mechanism, and light absorption












![2-[(Diphenylphosphoryl)methyl]phenol 70127-50-3 | TCI EUROPE N.V. 2-[(Diphenylphosphoryl)methyl]phenol 70127-50-3 | TCI EUROPE N.V.](https://www.tcichemicals.com/medias/H1763.jpg?context=bWFzdGVyfHJvb3R8MjYyMDN8aW1hZ2UvanBlZ3xoYTYvaDUxLzkxMzgwMDMyNzk5MDIvSDE3NjMuanBnfGU1ZmE3YWZmNmIzYzhlZjVjOTM0ODNjYTIyMjk0NWQzNGYyMDM1OTU5MjQyNGIyNGE5YjQwZDY2YmE2NWRlMzg)





